In addition, systemic administration of allogeneic MSC treatment in pediatric patients with acute GvHD showed a significant improvement in survival rate and a sustained therapeutic effect at 6 months after treatment (NCT02336230). 2018 saw the first marketing approval of an allogeneic MSC product for the local treatment of complex perianal fistulas in Crohn’s disease 11. This potent effect is achieved through the MSC secretome, a complex mixture of bioactive factors and extracellular vesicles (EVs) 10.Ĭlinical application of MSC technologies has been explored for some years and work is already underway. MSCs provide immunoregulatory assistance in order to quell this overactive Th1 profile and are a dynamic cell population that are apt to respond therapeutically to an inflamed cell population 7, 8, 9. A skew towards a pro-inflammatory Th1 phenotype has been implicated in aberrant cell-mediated inflammation and autoimmune response in diseases such as type 1 diabetes, rheumatoid arthritis, multiple sclerosis, and systemic lupus erythematosus 5, 6. T cell activation is essential to an effective immune response, but prolonged T cell activation can lead to chronic inflammation and immunopathology. 
MSCs represent a way to inhibit lymphocyte activation and reprogram the T and B cell sub-compartment through paracrine signaling mechanisms with a high degree of specificity. Several studies have shown the differentiation of monocytes into regulatory macrophages 1, 2, 3, dendritic and T cells into tolerance-inducing phenotypes 4, and even naïve B cells into plasma cell types. 
Mesenchymal stromal cells (MSCs) have been described by many as potent immunomodulators capable of reprogramming, or differentiating, immune cells. In complex conditions where single molecule immunotherapies have failed or cannot address broad, systemic pathways, cell therapy offers a compelling alternative approach. But immunotherapy can also be deployed to broadly modulate the immune system and/or bring homeostasis to a dysregulated immune response. Recently, cellular immunotherapy has drawn particular attention, especially in the field of oncology, as novel CAR-T therapies are being developed to elicit or amplify immune responses with tumor cytotoxicity as an end goal. Immunotherapy can be broadly described as treatment of disease through the modulation of the immune system. This coculture technology can have broad implications for use in studying MSC-immune interactions under flow conditions as well as in the generation of ex vivo derived immune cellular therapeutics. This platform was useful to study a model of immobilized MSCs and circulating immune cells and showed that monocytes play an important role in MSC driven immunomodulation. Furthermore, circulating, activated human peripheral blood mononuclear cells (PBMCs) were suppressed by MSC bioreactor culture confirmed by a durable change in their immunophenotype and function. Exposure of bioreactor-seeded MSCs to inflammatory stimuli reproducibly switched MSC secreted factor profiles and altered microvesicle composition. MSCs showed a stable and predictable metabolite and secreted factor profile during several days of perfusion culture. 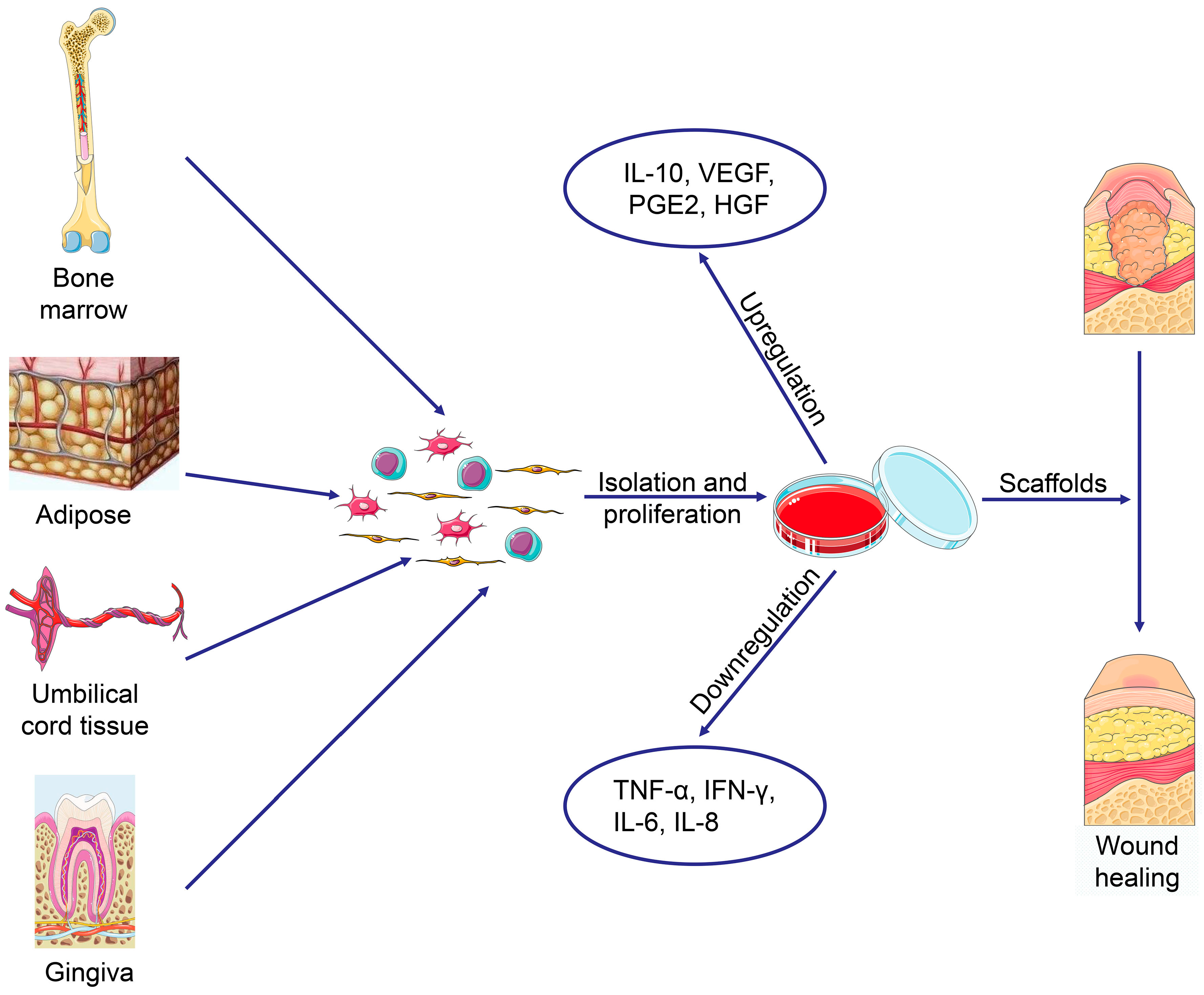
MSCs were seeded on the extraluminal side of hollow fibers within a bioreactor where they indirectly interact with immune cells flowing within the lumen of the fibers. Here, we report the ability of a scalable hollow fiber bioreactor to effectively maintain ideal MSC function as a single population while also being able to impart an immunoregulatory effect when cultured in tandem with an inflamed lymphocyte population. Clinically, they have shown some promise but with limited success. Bone marrow mesenchymal stromal cells (MSCs) have been studied for decades as potent immunomodulators. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
